How the Newest Diesel Engines Emit Very Little Greenhouse Gas Nitrous Oxide
Latest catalytic converters reduce pollution by unusual mechanism
(September 2013)
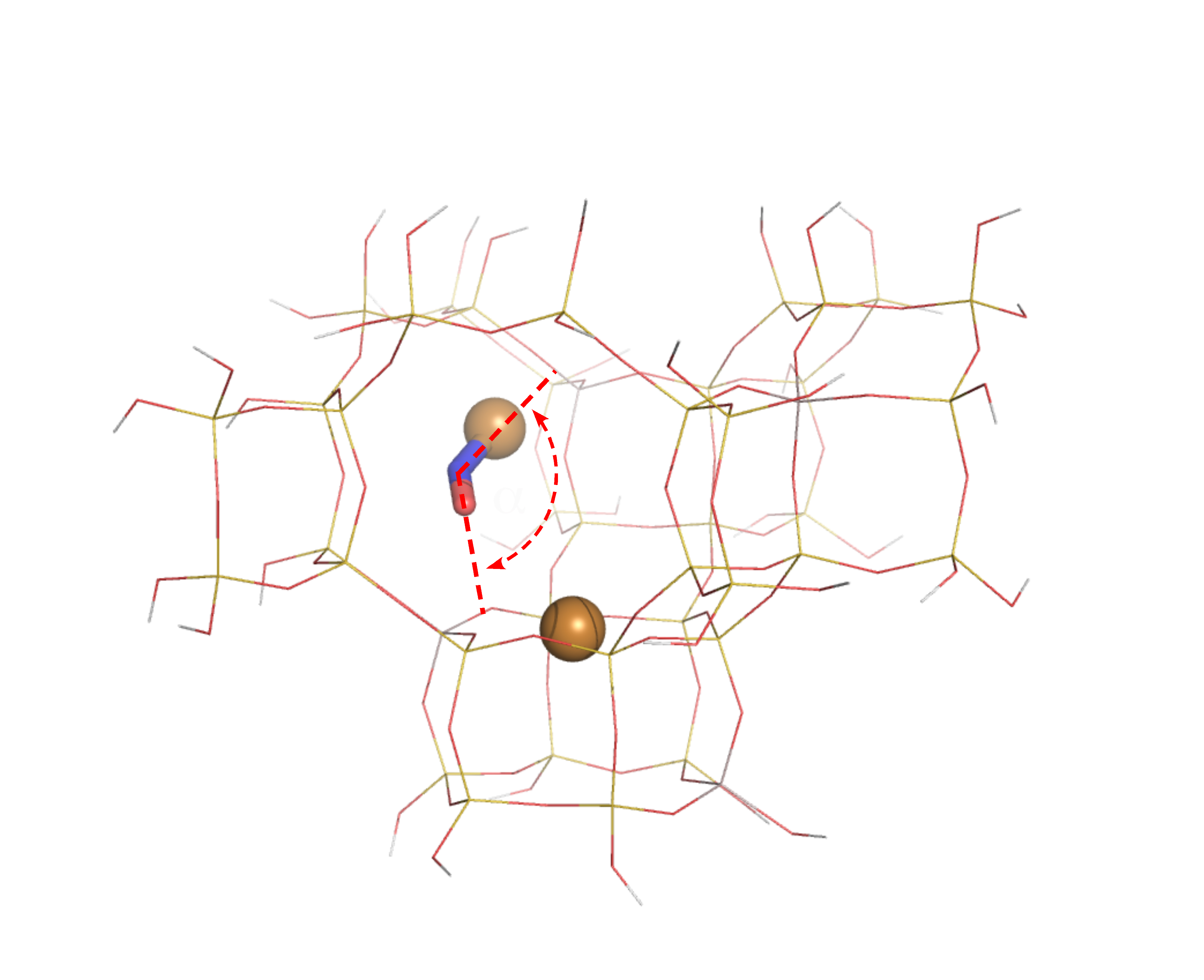
This computer model of a zeolite catalyst shows nitric oxide (ball-and-stick) interacting with a positively charged copper ion (copper ball) at an unexpected angle (red dotted lines). Photo courtesy of Kwak et al. Enlarge Image
Results: The newest catalytic converters in diesel engines blast away a pollutant from combustion with the help of ammonia. Common in European cars, the engines exhaust harmless nitrogen and water. How they do this hasn't been entirely clear.
Now, new research by scientists at Pacific Northwest National Laboratory shows that the catalyst attacks its target pollutant in an unusual way, providing insight into how to make the best catalytic converters.
Reporting in the journal Angewandte Chemie International Edition, a team of researchers in the Institute for Integrated Catalysis at PNNL showed that the artificial catalyst works much the same way that similar bacterial enzymes do: by coming at the target from the side rather than head on.
See more about this discovery, which was supported by the Department of Energy Office of Energy Efficiency and Renewable Energy, in the PNNL news release.
Reference: Kwak JH, JH Lee, SD Burton, AS Lipton, CHF Peden, and J Szanyi. 2013 "A Common Intermediate for N2 Formation in Enzymes and Zeolites: Side-On Cu-Nitrosyl Complexes." Angewandte Chemie International Edition 52(38):9985-9989. DOI: 10.1002/anie.201303498.
