Where There Is Waste, There Is Energy—and Opportunity
By John Holladay, Lynne Roeder, Bob Weber and Cynthia Jenks

Small, tailored mini-refineries located where waste is produced—such as farms and ranches, waste water treatment plants, food processing centers, and similar locations—could convert 85 percent of the nation's abandoned carbon into useful energy. By largely eliminating secondary processing and transportation costs, they could also reduce greenhouse gases by 30 percent.
Today's low energy prices feed both a false sense of security about U.S. energy availability and a continued appetite for carbon-based fuel and chemical sources. But as history shows, energy prices and oil imports will rise again, swinging the pendulum back toward concerns over energy security and domestic fuel alternatives.
At the same time, the U.S. has an inventory of largely abandoned carbon with an aggregate energy content equivalent to the amount of petroleum we import. These resources, which include low-energy-density wet waste and sludge; forest residue; agricultural, industrial and municipal waste; COX; and flare gas—are geographically dispersed and chemically diverse.
Small, tailored mini-refineries could take advantage of these abandoned resources, significantly change how we use carbon, and reduce environmental impacts. The small scale enables communities to couple low-value carbon resources with seasonal energy demands and other constraints with no offsite shipping of waste. Converting these unused resources may produce kerosene or chemical feed precursors depending on local communities' needs.
Though daunting process scaling challenges must first be overcome, regional networks of flexible, modular mini-refineries at farms and ranches, wastewater treatment plants, food processing centers, and similar locations could yield the following:
- Appropriate scaling for the resources and needs of the local community
- Regional economic growth and jobs
- Secure, stable fuel supplies for military installations
- Better waste management and water resource protection
- ~60 percent reduction in greenhouse gases compared to the fossil fuels and/or chemicals they replace.
Yet, there are challenges that will need to be met for progress to be made on this big idea in the next decade.
1Annual resource: Wet waste can be converted into the equivalent of 1.2 billion barrels of oil (BOE); agriculture residue and forest residues (resource priced <$60/tonne) not currently used account for 1 billion BOE; available Municipal solid waste accounts for 0.8 billion BOE per year and flared gas 0.04 billion BOE per year (EIA, EPA, ORNL, and PNNL)
Scaling—aye, there's the rub
With support from the Department of Energy, PNNL has partnered with industry to make significant advances in biomass processing research and development for three classes of stranded carbon:
- Gas – working with LanzaTech to convert carbon monoxide offgas from steel mill plants to make jet fuel
- Wet – working with Genifuel and the Water Energy Research Foundation on hydrothermal liquefaction to convert wet sludges—food waste, water treatment sludge, algae—into biocrude oil using a simple reactor configuration that Genifuel is moving to pilot scale
- Dry – working with DOE and Battelle to combine pyrolysis and hydrotreating to produce high-quality gasoline and improve reactor operability by a factor of 60, from 25 to 1500 hours.
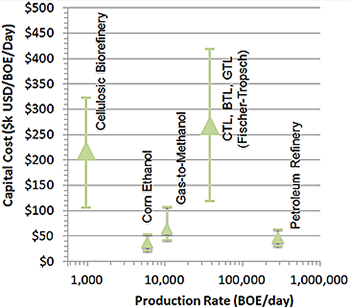
Fuel conversion technologies that scale up often do not economically scale down.
To realize these processing advances at small scales but with high production, two primary challenges must be overcome: 1) the limitations of current catalyst options that can break the "power law" associated with scaling, and 2) engineering for modularization and replication.
In designing for industrial operations, the "power law scaling" of capital costs typically drives conversion technologies to very large scales. Such plants can cost billions of dollars to construct and require continuous input of massive quantities of feedstocks. In addition, large-scale processes are usually designed to operate at severe conditions of pressure and temperature because 1) they can afford to employ 24/7 shifts of highly trained operators, and 2) they can find a profitable sink for high-quality heat (e.g., cogeneration of power, thorough heat integration). The severe reaction conditions mitigate catalyst poisoning and deactivation. Therefore, conventionally scaled plants don't fit the regional, distributed nature of the lowest cost feedstocks targeted here. Nor are such plants amenable to operation near the feedstocks, where communities may not welcome industrial-scale facilities but might tolerate smaller scale, distributed plants.
We see two classes of impediments to implementing the distributable plants:
Engineering. Separation and conversion platforms must be tied to reactor engineering and process intensification at scales that allow small modular units to be economically attractive. Advances in monitoring and control systems will be needed to enable turnkey operation and minimize staffing—important factors for small or remote communities.
Catalysis. Hydrotreating accounts for over half the capital and 85% of the operating cost (not counting feedstock) for converting bio-oils to gasoline and diesel fuel. A need remains for more active, more stable catalysts that operate in water with bioderived materials containing sulfur, nitrogen, and alkali. And we need to understand the mechanism for catalyst inhibition and reactor fouling from sugars and derivatives. Understanding these mechanisms and developing mild regeneration techniques are critical for moving new technologies into industrial use.
We are also examining other ways to remove oxygen without impacting carbon yield, such as electrocatalysis. If we can couple this method with more active acid-base catalysts, we can find new ways to produce higher quality diesel fuel and gasoline at lower costs and smaller scales that match distributed carbon.
Small-scale footprint, wide-ranging applications
Building on advances in catalysis science for biofuels applications, DOE's Bioenergy Technologies Office is supporting a workshop in December co-organized by Ames Lab, PNNL and Iowa State University, to explore modular mini-biorefinery concepts, including the catalysis technologies suitable for small scales. In fact, scientists from 12 national laboratories are working together to define and come up with new solutions to the problem. At PNNL, we are applying our analysis tools to help understand the logistics of managing the waste feedstock concept. With the national labs' remarkable and unique capabilities, DOE can facilitate cooperation with key university and industrial partners and stakeholders. The Institute for Integrated Catalysis is front and center in applying fundamental science to most challenging catalyst questions.
Key stakeholders that need to be engaged include waste producers, such as state/local municipalities; ranches/dairies; food processing plants; waste management companies; and the chemical industry. Others may include depots, harbors, railroads, marine, off-road construction, and mining. Finally, groups such as non-government organizations, small business administrations, and underwriter laboratories could be interested.
DOE has among the strongest conversion programs in the U.S. and is best suited to take on this leading effort and integrate with NSF, USDA, DOT, DOD, EPA, and Commerce, as well as international agencies. A highly focused effort, bringing together related work in EERE, FE, ARPA-E, and SC, along with key stakeholders, will make possible the incorporation of mini-refineries that can scale down to 2 to 200 barrels/day production at the very low-capital thresholds required to enable rapid market adoption.
John Holladay is associate director of the Institute for Integrated Catalysis at Pacific Northwest National Laboratory, Lynne Roeder is a senior specialist for PNNL's Energy and Environment Directorate, Bob Weber leads research activities in heterogeneous catalysis for the IIC, and Cynthia Jenks is assistant director for scientific planning and division director of chemical and biological sciences at Ames Laboratory.

