The Fine Arts of Catalysis and Consensus
Interdisciplinary team fundamentally shifts our view of catalytic anchors on metal-supported graphene
"There was vigorous agreement between the theory and experiments," said Dr. Roger Rousseau, who leads theoretical chemists at Pacific Northwest National Laboratory. "But agreeing never stopped us from arguing." And that was the tone of the interdisciplinary team that worked through years of research to upend the long-held view of how oxygen atoms bind on metal-supported graphene sheets.
"How the oxygen binds is different from what many others think, and we were able to provide the evidence for it," said Dr. Vanda Glezakou, a PNNL theorist on the study.
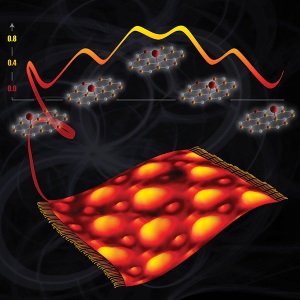
The team began with a graphene sheet thrown like a carpet over ruthenium metal. Oxygen atoms settled on the wavy surface. The experimentalists heated the sample inside a scanning tunneling microscope. The oxygen atoms started hopping, and from how the atoms moved, the team determined precisely where and how the oxygens were bound. The microscope did not have enough resolution to simply image where the oxygen atoms sit relative to the lattice of carbon atoms that make up the graphene sheet and the ruthenium support, so this approach was used.
For months and months, experimentalists working in DOE's Environmental Molecular Sciences Laboratory, an Office of Science user facility, measured both the distance and the direction that the oxygen atoms hopped. But it wasn't enough. They needed additional insights from theory and calculations.
Having theorists and experimentalists work closely together might be unusual at some institutions, but not for these catalysis researchers. "This is normal," said experiment lead Dr. Zdenek Dohnálek, who holds a joint appointment with PNNL and Washington State University. "It is both beneficial and challenging for us all."
From a theory standpoint, the problem was complex. They needed to re-create the sample that was in the microscope inside a computer simulation. The size of the metal lattice and the number of electrons that had to be included were daunting. "The necessary calculations were backbreaking in terms of processing time, and the data analysis had to be clever," said Rousseau of the model that was used in the 700-atom calculations. Completing the calculations required resources at the National Energy Research Scientific Computing Center, another DOE Office of Science user facility.
After running the calculations, the theorists and experimentalists would get back together to discuss the findings. Often, the discussions led to new experiments, new analyses, and new simulations. As the effort progressed, the team started to see how their work was about to change the conventional wisdom that a single oxygen atom nestles between two carbon atoms, sharing bonds with both carbons on metal-supported graphene.
Ultimately, the team's persistence paid off with a detailed understanding of why oxygen hops across the hot surface and how it moves. They determined exactly how oxygen atoms bind and how the energy changes as they move across different sites on the surface. Specifically, oxygen atoms bind preferentially on top of carbon atoms that are closer to the underlying ruthenium, while binding over two carbon atoms is less preferred; binding over graphene that is far away for the ruthenium surface is even less favorable. This behavior makes oxygen atoms hop two carbon sites at a time as they dash across the graphene surface.
Having answered how oxygen behaves on carbon, the team is planning to use the oxygen as anchors to tether single metal atoms and small oxide clusters, and build model catalysts.
Sponsors: Department of Energy, Office of Science, Office of Basic Energy Sciences, Division of Chemical Sciences, Geosciences, and Biosciences (Z.N., M.T.N., V.A.G, R.R., Z.D.); an Alternate Sponsored Fellowship at Pacific Northwest National Laboratory (F.P.N.); and the University of Graz (F.P.N.)
References
Novotny Z, MT Nguyen, FP Netzer, VA Glezakou, R Rousseau, and Z Dohnálek. 2018. "Formation of Supported Graphene Oxide: Evidence for Enolate Species." Journal of the American Chemical Society. DOI: 10.1021/jacs.7b12791

